Pharma’s Sputnik Moment
Mutually assured drug discovery
“I’m really hoping your industry moves from drug discovery,” Jensen Huang quipped, “which is kind of like wandering around the forest looking for truffles.”
The other guy laughed, nodded, and by the time the session ended, agreed to commit a billion dollars over five years to Nvidia’s stack — talent, infrastructure, and compute. The two men shook hands.
The other guy was David Ricks, the chief executive of Eli Lilly — the most valuable pharmaceutical company in the world, a $700 billion firm built over a century, responsible for some ho-hum compounds like insulin, Prozac, and Zepbound. Ricks and Huang had been on a stage at the JPMorgan Healthcare Conference in San Francisco, widely regarded as the most important annual gathering in pharma.
These were two men, among the most important in their fields, which also happen to be two of the most important industries on earth.
The relationship has a hierarchy. There is a speed differential. There is an incumbent and an insurgent. And the insurgent is becoming a real threat to the incumbent.
Pharma is slow; AI labs are fast. Pharma is regulated; AI labs are not (as much). Pharma still believes it sets the terms of its own industry; AI labs already know it does not. Everyone in the room knew the hierarchy. Huang said it in a sentence, with a joke, and Ricks laughed along. Two men, one check.
Eli Lilly had just bought ten million truffles.
“A century of human progress in science could be achieved by AI in just 5 or 10 years.”
Dario Amodei had said it from a stage at Davos a year earlier. Three months after the JPMorgan session, Anthropic quietly acquired the startup Coefficient Bio for $400m in stock. Coefficient was only eight months old, and had fewer than ten employees. It had no wet lab, no clinical capacity, no Investigational New Drug (IND) application. What it boasted was a small team that had come out of Genentech’s machine-learning brain trust, Prescient Design.
Anthropic is not the only foundation lab on the prowl. Within two weeks of the Coefficient deal, OpenAI announced its own pharmaceutical partnership with Novo Nordisk, and just two days later, launched GPT-Rosalind, a biology research agent built specifically for drug discovery workflows. Earlier in the year, Google’s Isomorphic Labs announced a research collaboration with Johnson & Johnson and now expects its first AI-designed compounds to enter clinical trials by the end of 2026. Every major frontier lab is making the same bet at the same time.
I’ve spent the last several months trying to explain to friends in tech why the frontier labs are racing into pharma, and why pharma — the industry that fifteen years ago would have responded to a GPU seller with a polite rejection email — is responding now with billion-dollar contracts and stage time at its most important annual conference.
Pharma is having its Sputnik moment.
The reason this matters past the boundaries of either industry is that the relationship being drawn between AI and pharma right now will determine, over the next decade, how fast new drugs reach patients, which diseases get pursued and which do not, and how much of that calculus happens inside companies whose decisions you can see versus inside companies whose decisions you cannot. It is the story of the system that decides whether the drug your father needs gets approved in three years or twelve.
For most of the last century, pharma was a unipolar power inside drug development. It set the timelines. It set the pace. There was no peer pressure. There was nothing upstream of it that could move faster, no force pulling it toward urgency.
That has now changed. The frontier labs are not coming for pharma’s manufacturing or its trials or its distribution — they cannot, and I will get to why. They are coming for the part of the work that compounds in software, and they are moving at a speed pharma has never had to compete against. This is not a takeover. It is a forced partnership — closer, faster, and stranger than either industry has been in before. Two powers, neither able to fully absorb the other, each forced by the other’s existence to move faster than it would on its own. The space race was not produced by NASA alone. It was produced by NASA and the Soviets, in a relationship neither side wanted but both sides operationalized.
AI is not going to eat pharma. Pharma is not dying. But truffle jokes and biotech acquisitions are the same story told from two ends — in public, and on the cap table.
What are AI labs getting out of this?
The press releases call it partnership. The honest answer has something to do with pharma being a $1.5 trillion industry whose entire upstream layer — the part that runs on reasoning, the part the labs can do — is suddenly available as a recurring, high-margin software workload at a scale comparable to all of frontier AI’s existing revenue combined. That is the first thing the labs are after.
The second is data nobody else can buy. Pharma owns the world’s most valuable scientific corpus: molecular libraries, target validation experiments, decades of trial outcomes, the priceless record of which compounds failed and why. The labs are paying to train on what pharma knows.
The third is justification. A frontier lab cannot defend $100 billion in capital expenditure on coding. It can defend that expenditure on curing disease. Biology is the second hard-science vertical, after software, where the labs can credibly say their models do real scientific work — and drug discovery is the most legible version of biology. It’s a rare PR win for the AI labs to say, “we’re curing cancer” as opposed to “we’re building AGI, and it may or may not ruin your lives”

The fourth is lock-in. If a lab’s reasoning model becomes the default upstream layer for a pharma company’s R&D, the switching costs are enormous. This is the AWS play. The labs are buying the floor on which the market stands.
YOU HAVE most certainly heard a version of this story before. The “century of biology.” AI eats pharma. Sam Altman has said it. Demis Hassabis has said it. The cumulative payoff, after a decade, has been modest — more than 170 AI-discovered drug programs are in clinical development as of early 2026. None have completed a Phase III trial.
What’s different now is that the frontier labs have stopped trying to sell pharma a tool. They are now infrastructure that pharma rents — and pharma, having signed the contracts, is now in a co-dependent relationship it cannot easily leave.
January was the moment that relationship stopped being subtle.
Let’s look at what the labs are buying, and what they are not.
Anthropic’s pharmaceutical partners include a catalogue of major drug sponsors: AstraZeneca, Sanofi, Genmab, Novo Nordisk, AbbVie, Flatiron Health. OpenAI’s are similar: Amgen, Moderna, Thermo Fisher, the Allen Institute, Dyno Therapeutics. In April, Novo Nordisk and OpenAI announced a tie-up that their press release described as covering everything “from drug discovery to commercial operations,” with full integration by year’s end. Lilly committed $2.75 billion to Insilico Medicine in March and a billion to Nvidia in January. GPT-Rosalind, OpenAI’s biology research agent, launched April 16.
Each deal is slightly different, but the baseline is that the lab provides the model, while the drug company provides the asset, the trial sites, the regulatory effort, and, eventually, the patients.
What no AI lab has bought is any actor in the actual drug development process. There is no lab acquisition of a contract research organization (CRO). There is no lab acquisition of a manufacturing site. There is no lab acquisition of a clinical operations team. What the labs are acquiring is computational biology talent. Marc Tessier-Lavigne, formerly Genentech’s chief scientific officer, is now running Xaira Therapeutics, with $1billion in committed funding. Arvind Rajpal, who ran Genentech’s large-molecule discovery group, went with him. Kyunghyun Cho, one of the formative figures in modern machine learning and the architect of Genentech’s internal AI effort, left for NYU in January. The team he built was already gone — Nathaniel Stanton and Andrew Frey among them — absorbed into Coefficient, then into Anthropic.
IF YOU want to see the shape of the next five years in pharma, you should pay attention to the asymmetry in the deals, not just the deal sizes.
AI labs have taken the half of pharma that scales like software. Pharma has kept the half that does not. And this is where we get our Cold War equilibrium.
The half that scales like software is everything upstream of human contact: target identification, hypothesis generation, molecular design, regulatory document drafting, trial protocol simulation. The half that does not scale like software is everything that involves a human body in regulated space: enrolling patients, running trials across forty sites in twelve countries, manufacturing biologics under FDA inspection, navigating the eighteen months between a Phase III readout and a label. The first half has a marginal cost approaching zero. The second half is where Eroom’s Law (the inversion of Moore’s Law) applies, causing pharmaceutical R&D productivity decline by a factor of 80 since 1950. It now costs roughly $2.5 billion to bring a drug to market, against $40 million in inflation-adjusted dollars when Paul Janssen was setting up his lab in 1953.
Couldn’t the labs, in principle, acquire the rest of the stack? Couldn’t Anthropic — with a possible $1 trillion IPO looming — buy IQVIA? Couldn’t OpenAI, larger still, buy Charles River, or Catalent, or one of the big CROs, and finally close the loop? Wouldn’t it be more profitable to compete with pharma all the way down to the patient instead of partnering with it?
Well, no.
IQVIA has eighty-eight thousand employees and a market capitalization around $40 billion. ICON has forty-two thousand and is worth $15 billion. Charles River, Parexel, Lonza, Samsung Biologics, WuXi: collectively have several hundred thousand people working in regulated, relationship-driven, decades-old institutions whose entire competitive moat is precisely that they cannot be compressed into a foundation model. Catalent, the largest contract manufacturer in the West, was bought off the public market by Novo Holdings in 2024 for $16.5 billion and is now off the table. The academic medical centers that run the most important oncology trials in this country — MD Anderson, Memorial Sloan Kettering, Mass General — are nonprofits (you cannot acquire a nonprofit). And you cannot run a Phase III trial across forty sites in twelve countries from a foundation model. The work is done by humans, in regulated buildings, on a timeline the FDA enforces, in relationships that took thirty years to build.
The labs could not absorb this if they tried. The cap tables are upside down — Anthropic and OpenAI are now larger than every pharma company except Lilly, but their valuations are paper, denominated in optionality rather than cash, and the hundreds of thousands of trained clinical operators they would need to hire do not exist outside the existing system. Even if the math worked, the strategic logic does not. A frontier lab buying a CRO would not capture pharma’s execution layer; it would simply become a worse, more diluted version of it — a two-thousand-person research lab trying to absorb a fifty-thousand-person regulated services business inside one balance sheet. The two operating models are not compatible. Software margins do not survive contact with clinical operations.
That is the line in the concrete. The labs will take everything that compounds in software — but they will reach a stop, because the next layer down starts compounding in human bodies, regulated space, and the slow accretion of trust between a sponsor and a site coordinator who has been running enrollment for twenty years. That is the partition. The frontier labs are not displacing pharma from execution. They can’t. They are displacing pharma from sovereignty. The upstream layer of pharma’s work — the hypothesis generation, the molecular reasoning, the literature synthesis, eventually the regulatory drafting — will from now on run through five companies. Pharma will negotiate with them the way retailers negotiate with AWS. The negotiation will go about as well.
Isomorphic Labs, the Alphabet/DeepMind spinout, is the closest thing to a caveat. The company has roughly $3 billion in combined deal value across separate partnerships with Novartis and Lilly, $600 million in primary capital from Thrive, and an oncology pipeline approaching trials. Demis Hassabis said at Davos this January that the first AI-designed Isomorphic drugs would enter trials by the end of 2026 — a year behind the original target. When they do, they will run on Novartis and Lilly infrastructure. Isomorphic’s stated plan is to license programs out at later stages. They design internally and rent execution, for now. The partition still holds.
WHAT DOES this mean for someone outside the industry?
If your cousin has a rare cancer and her drug has been stuck in Phase II since 2022, that wait may finally start to shrink. If your father’s Alzheimer’s trial keeps missing its readout date, an answer might be close.
This might be the best thing that has happened to drug development in over fifty years. The unipolar century did not produce drugs at the pace it could have. Pharma’s slowness was treated as a fact of nature because there was nothing in the world that could pressure it to be otherwise. It is now being forced to accelerate because of visibility. When two industries are always on the same stages, the same press releases, and the same partnership announcements, the difference in their pace becomes glaringly clear. Pharma is now next to a younger, energetic counterpart that runs three orders of magnitude faster, and pharma has become the obvious bottleneck. The pressure is on.
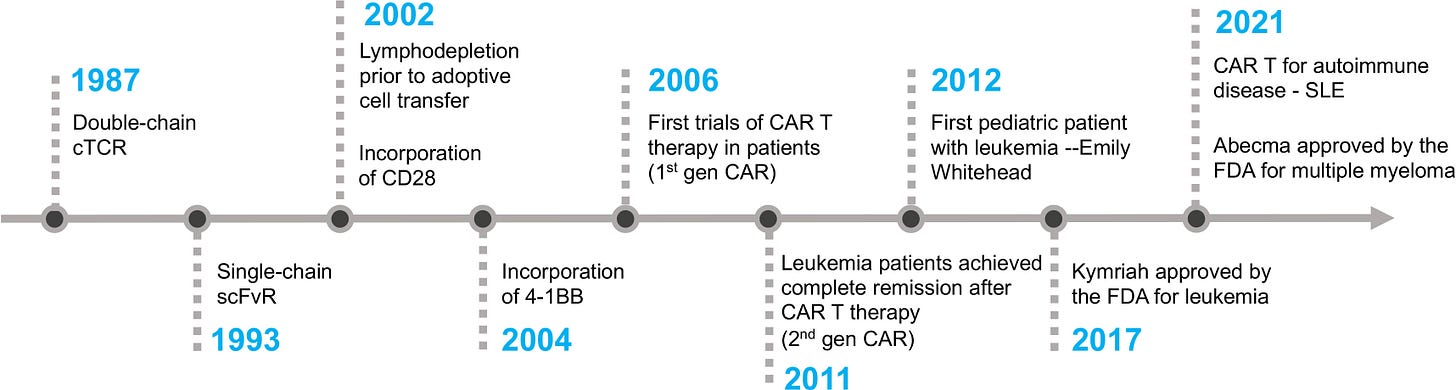
Some of the things patient advocates have been pushing for thirty years — adaptive trial designs, better endpoints, faster regulatory review for accelerated-approval candidates — are already on the page in 2026 FDA guidance. Speed is contagious. The Soviets did not invent the transistor, but they made America build the moon program. Most of what was scientifically extraordinary about the second half of the twentieth century came from the competition, not from either pole acting alone.
There is also a real worry, which I think of in three clusters.
The first is opacity that comes with consolidation. The upstream layer of a regulated, life-or-death industry is moving into a small number of private companies whose contracts are confidential and have looser obligations to disclose. None of the major partnership announcements this year has been accompanied by financial terms or anything resembling specific governance language. When Novo Nordisk announced its partnership with OpenAI in April, the phrase “strict data governance” appeared once, as a bullet at the top of the press release, and was never defined again. The deal may be perfectly reasonable, but we wouldn’t be able to tell.
The second is target selection. Pharma has always decided which diseases, which proteins, which patient populations get pursued. A drug company prioritizes targets where it can defend a patent, run a tractable trial, and price into a paying market. Those incentives produce problems (rare diseases get neglected, common diseases in poor countries get neglected more), but you can name them. When the reasoning layer concentrates in five labs whose business model is built around dataset reuse and broad applicability, the prioritization criteria change shape. A frontier lab is rewarded for designing a molecule that works across many indications, that generates training-quality failure data, that supports its next pharma partnership.
The third is concentration of failure. The unipolar century distributed pharma’s bad bets across dozens of companies running independent pipelines; if Pfizer’s CNS group missed something, Roche’s CNS group might catch it. The bilateral structure threads everything through five models. If GPT-Rosalind has a blind spot in how it reasons about, say, blood-brain barrier permeability, every drug company that uses the model simultaneously inherits the same blind spot.
All things considered, pharma is now left with two plausible plays. It cannot build the labs in-house — the talent is gone and the ship has sailed. It cannot buy them — no pharma cap table is large enough to move the needle at Anthropic. It can rent, which it already does, and it can aggressively compete on the half it still owns: trial execution, regulatory navigation, manufacturing, distribution. The second one is where the next ten years get interesting.
THE PRODUCTS of the Cold War – the moon program, the integrated circuit, federal science funding — were the result of two nations pushing each other to the limits of what was humanly possible. That relationship, however, was always held at a geopolitical distance.
The pharma-AI relationship has the same kind of push and none of the distance. This time, the pressure is coming from inside the same country, the same cap tables, the same regulatory system, the same conferences. Nvidia and Lilly were on the same stage in San Francisco in January. Their CEOs shook hands and signed a billion-dollar contract in the same afternoon. The Soviets and the Americans had backchannels, treaties, and the occasional handshake. They never had a billion-dollar contract.
What the next five years will produce is warmer and closer than any Cold War rerun. The Cold War’s compressions were built across a border, between governments that did not share a language or a currency. The pharma-AI relationship shares a country, a regulator, and a drug-development pipeline that needs both halves to function.
If the distanced version of this dynamic produced the second half of the 20th century, the proximity version will dwarf it. The sick cousin in Phase II since 2022, the father waiting on an Alzheimer’s readout — their timelines depend on a relationship that is going to be more entangled than the one that put a man on the moon.
White Alba truffles still sell for six thousand dollars a pound, and the best ones still come from foragers in Italy who have spent twenty years learning specific groves. The forests are the same forests. It comes down to who owns the tools, the talent, and the knowledge.










https://centuryofbio.com/p/sid the GitLab Founder went “founder mode” on his own deadly spinal bone cancer
Good article, but why no mention of practical recent use of AI to develop successful cures (therapeutics? vaccines?) both in Australia (ie AI engineer saves his dog from Mast Cell Cancer) and USA (ie GitLab Founder saves his life from Spinal Bone Cancer)? 🤔🤔🤔